Thermal stability of β-H2CO3 in the product mixture at 260 K, ∼400 mbar... | Download Scientific Diagram

Sejumlah H2CO3 (Ka = 4,3 x 1O-7) dicampurkan dengan larutan Ca(OH)2 membentuk larutan penyangga. Setelah - Brainly.co.id

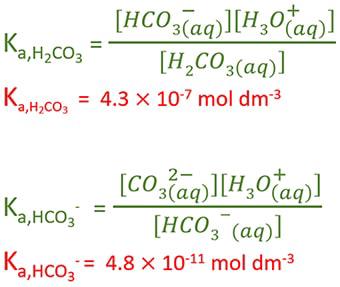
OneClass: Complete the Ka1 expression for H2CO3 in an aqueous solution. Do not include states in the ...
Find the concentration of H+, HCO^-3 and CO^2-3, in a 0.01M solution of carbonic acid if the - Sarthaks eConnect | Largest Online Education Community

SOLVED: If the carbonic acid (H2CO3) concentration in a sample of blood is 0.00125 M, determine the bicarbonate ion (HCO3-) concentration required to buffer the pH of blood at pH = 7.40.
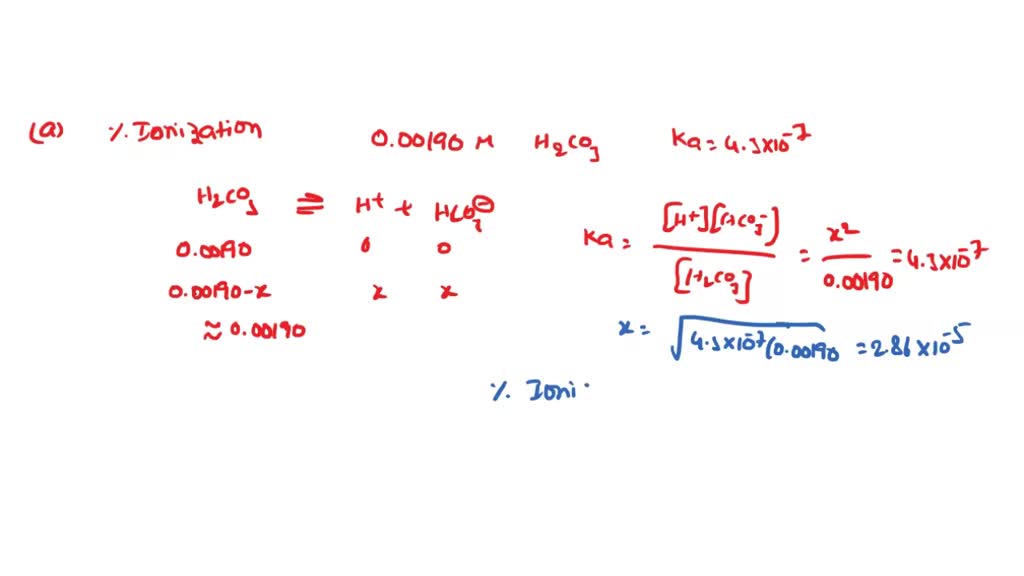
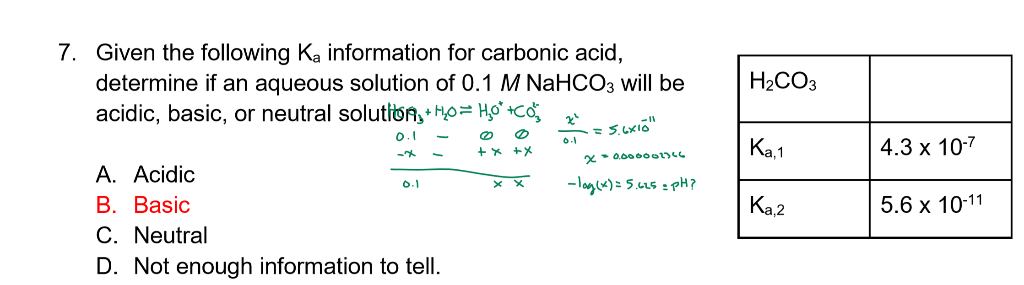
SOLVED: Calculate the percent ionization of carbonic acid (H2CO3) in solutions of each of the following concentrations (Ka = 4.3e-07.) (a) 0.281 M % (b) 0.366 M % (c) 0.641 M %
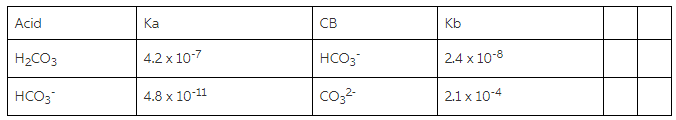
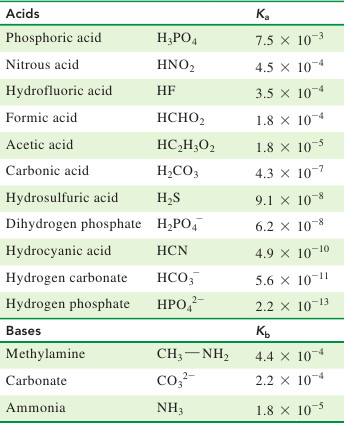
A) Using the information in the chart about Ka/Kb values, whichspecies will you use? - Home Work Help - Learn CBSE Forum